COVID-19 and Antimicrobial Resistance (AMR) and Pandemics
8/5/2020

Written by:
Romney M. Humphries, Vanderbilt University Medical Center, Nashville, TN
Graeme Forrest, Oregon Health and Science University, Portland, OR
Janet A. Hindler, Los Angeles County Department of Health, Los Angeles, CA
As the world grapples with the COVID-19 pandemic, many lessons are being learned, not the least of which is how to better prepare for future pandemics. This includes addressing the ongoing antimicrobial resistance (AMR) pandemic, which at a global scale has the potential to eclipse COVID-19. It is estimated hundreds of thousands of patients die globally each year due to complications associated with AMR infections—some projections indicate this number will be 10 million by 2050.1 In Part I of this feature, we provide information about use of antimicrobials and AMR during COVID-19. We explore how COVID-19 has impacted one hospital’s antimicrobial stewardship program. In Part II, we discuss the ongoing AMR predicament and what clinical microbiologists can and should do now, based in part on knowledge acquired by clinicians, researchers and public health professionals from COVID-19.
Part I. What Do we Know About Antimicrobial Resistance (AMR) During COVID-19?
COVID-19 is a disease caused by a virus and characterized by cough, fever and shortness of breath. For some patients, particularly the elderly and those with underlying conditions, infection progresses to severe disease, including acute respiratory distress syndrome. These patients often require mechanical ventilation and intensive care hospitalization. Antimicrobials are administered to approximately 75% of COVID-19 patients,2 many of whom receive broad-spectrum therapy.3 While at first blush, use of antimicrobials to treat a viral infection seems counter to antimicrobial stewardship efforts, antimicrobial use is supported by national guidelines due to presumed or confirmed bacterial co-infections. Although antimicrobial stewardship is in full force, clinicians may continue antimicrobial therapy when faced with severely ill patients even if bacterial infection cannot be confirmed. One hospital’s experience details these challenges, in the Case Study on page 8.
In past viral pandemics (ie, caused by influenza), secondary bacterial pneumonia was a major cause of mortality, affecting roughly 35-50% of patients.4 In contrast, secondary bacterial infections are rare for COVID-19. A meta-analysis of nine published studies documented 8% of patients hospitalized for COVID-19 experienced bacterial or fungal co-infections.2 Rates of co-infection ranged from 0–27% across the studies.3 The reasons for this variability are unclear, but may relate to differences in treatment practices (eg, use of corticosteroids), infection control policies and/or local epidemiology.3 Additionally, case definitions may play a role. Anecdotal reports from hospitals across the US indicate specimen collection (in particular of respiratory secretions) from patients with COVID-19 may not be performed due to infection control risk, meaning cases of co-infection often remain unconfirmed by the laboratory.
At a higher level, the COVID-19 pandemic has the potential to detrimentally impact global rates of drug-resistant infections.5 Some factors of the COVID-19 pandemic that could contribute to an increase in AMR include:
- Widespread use of antimicrobials for patients with COVID-19 for presumptive or confirmed secondary infection.
- A large new population of patients at risk for AMR infections due to use of mechanical ventilation and prolonged hospitalization. These patients would not traditionally be considered at risk for AMR, as they have had limited prior healthcare exposure.
- Potential lapses in institutional infection control and antimicrobial stewardship practices while institutions adapt to challenges of the pandemic.
- Depleted availability of personal protective equipment (PPE) which may reduce use for other infections that would normally require PPE.
- Use of contact precautions may be limited due to cohorting based on COVID-19 status versus other infections.
- Closure or limited access to ancillary clinics in favor of telemedicine, driving empiric antimicrobial prescriptions versus those prescribed following diagnostic testing.
- Individuals in the community resorting to use of leftover antimicrobial prescriptions in their medicine cabinet to self-treat.
The news is not all bad. Increased vigilance for hand hygiene, other infection prevention activities and continued efforts to educate the public on the appropriate use of antimicrobials may offset some of the above factors. Many healthcare facilities are reviewing or enhancing their strategies for addressing AMR concerns during COVID-19. A few examples include:
- Monitoring AMR trends in isolates from COVID-19 patients to inform empiric therapy needs at the local level.
- Adopting rapid diagnostics (eg, respiratory panels for agents other than SARS-CoV-2).
- Continuing to emphasize best practices for work-up of respiratory specimens (see ORWG News Update Volume 3 Issue 1 Winter 2018).
- Strictly enforcing recommended infection control practices for all patients, staff, and visitors in all healthcare facility locations.
- Ramping up assessment of additional practices that might be implemented to help control transmission of all types of microbes.
Clinicians, microbiology laboratorians, infection preventionists, and public health practitioners must be vigilant in monitoring the potential impact of these factors on the emergence of AMR in individual patients, as well as at institutional and regional levels. Preparedness activities, listed in Part II, can aid the laboratory in addressing the potential challenges associated with changes to AMR related to COVID-19.
References
1 O’Neill J. Tackling Drug-resistant infections globally: final report and recommendations. Review on antimicrobial resistance, Vol. London, United Kingdom: Wellcome Trust, 2016.
2 Rawson TM, Moore LSP, Zhu N, et al. Bacterial and fungal co-infection in individuals with coronavirus: A rapid review to support COVID-19 antimicrobial prescribing. Clin Infect Dis. 2020;10.1093/cid/ciaa530.
3 Clancy CJ, Nguyen MH. COVID-19, superinfections and antimicrobial development: What can we expect? Clin Infect Dis. 2020;10.1093/cid/ciaa524.
4 Morens DM, Taubenberger JK, Fauci AS. Predominant role of bacterial pneumonia as a cause of death in pandemic influenza: implications for pandemic influenza preparedness. J Infect Dis. 2008;198:962-70.
5 Rawson TM, Moore LSP, Castro-Sanchez E, et al. COVID-19 and the potential long-term impact on antimicrobial resistance. J Antimicrob Chemother. 2020;10.1093/jac/dkaa194.
One Hospital’s Reality in the COVID-19 Era
As our hospital fights COVID-19, we have faced several diagnostic and antimicrobial stewardship challenges. COVID-19 infections can rapidly progress from mild respiratory illness to overwhelming pneumonia with sepsis requiring intensive care unit (ICU) monitoring with ventilatory support. While caused by a virus, COVID-19 disease includes pathophysiologic features that are traditional hallmarks of bacterial infection: pneumonia on imaging; increasing C-reactive protein and procalcitonin levels; and elevated peripheral white blood cell counts. This dynamic has resulted in our clinicians using antimicrobials broadly to treat COVID-19 patients.
One early decision was an effort to reduce unnecessary laboratory testing as our hospital prepared for testing surges associated with the pandemic. Among the tests discontinued was routine methicillin-resistant Staphylococcus aureus (MRSA) screening for patients prior to admission into the ICU. Our site has a low rate of MRSA infections, and knowledge of a patient’s MRSA status through this testing normally resulted in rapid vancomycin de-escalation. Subsequent to eliminating these MRSA screens, vancomycin usage increased by over 50% in the ICU.
The broad-spectrum antimicrobials routinely given to severely ill COVID-19 patients in our hospitals include coverage for Pseudomonas aeruginosa. Such treatment was often excessive, as the majority of COVID-19 patients have no risk factors for Pseudomonas infection. Compounding this stewardship challenge is the fact that no change in therapy was considered, even after a COVID-19 diagnosis was rendered and bacterial cultures were negative. Markers normally used to help with antimicrobial de-escalation discussions, such as procalcitonin, were elevated in these patients, making de-escalation decisions even more challenging.
Finally, during the pandemic we have noted increases in the daily numbers of sputum, endotracheal aspirates and blood cultures submitted to the laboratory. These specimens were collected in response to patients who developed high fever and shock, which again reflect the disease pathogenicity rather than “bacterial super-infection”. Compounding this, as the number of days during which the patient was intubated increased and the patient did not respond to therapy, more cultures were sent with the hope of identifying the reason for the patient’s continued decline. Unsurprisingly, these cultures often yielded organisms commonly associated with colonization of the respiratory tract during mechanical ventilation, such as Stenotrophomonas maltophilia or P. aeruginosa. Because of the patient’s symptoms, these were treated as pathogens. By late in the patient’s hospitalization, the impact of antimicrobial usage could be seen to drive AMR in these bacteria. These have been unprecedented times and a lack of data available early on to assist management combined with the severe clinical condition resulted in pressure of testing and antimicrobial usage. When this is eventually over, examining the practices that could reduce overcollection of specimens and treatment with broad-spectrum antimicrobials could be implemented.
Part II. Preparing for an AMR Pandemic – the Role of the Clinical Microbiology Laboratory
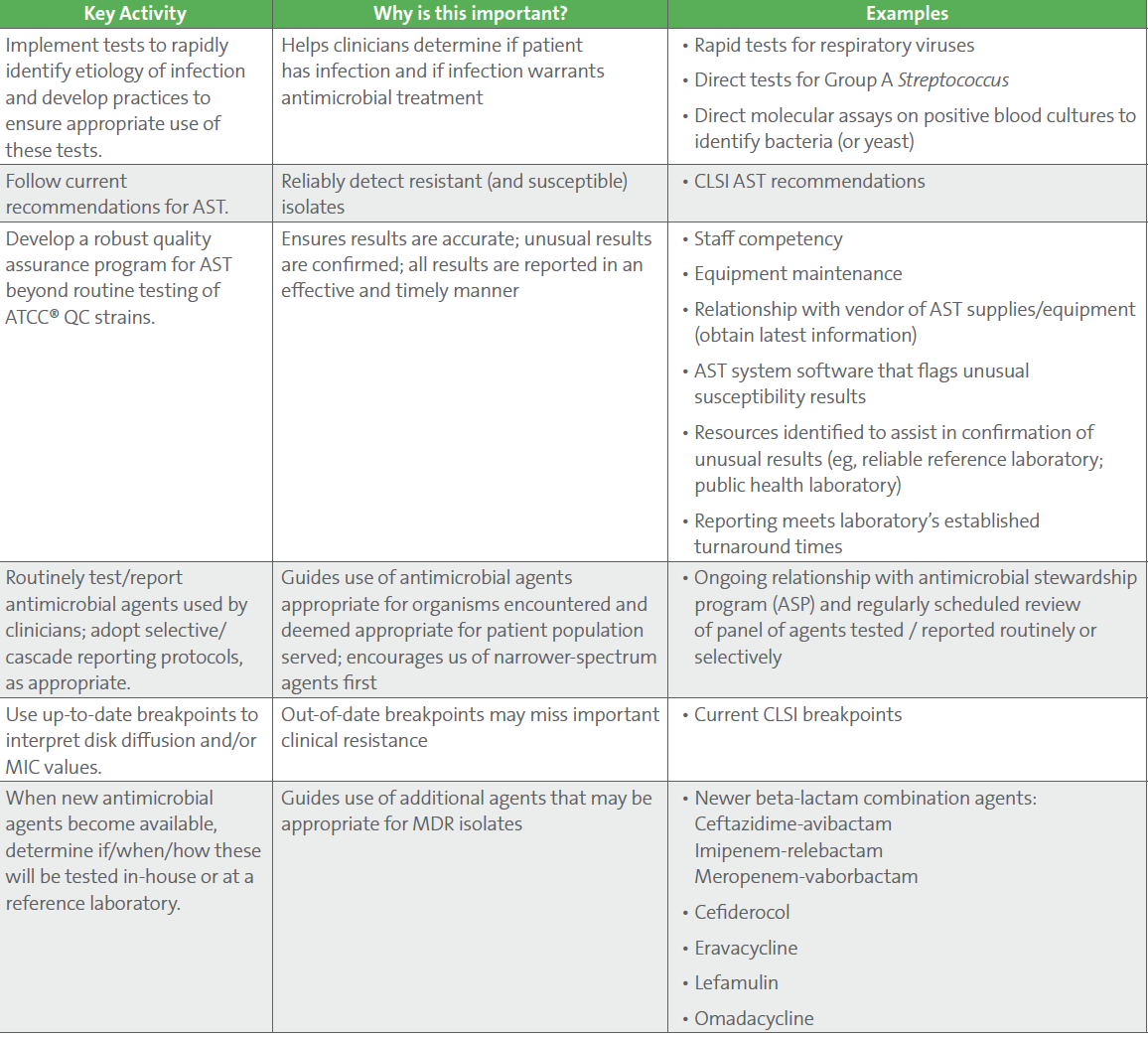
A major lesson learned from COVID-19 is the critical role of laboratory testing, and surge testing capabilities for crisis management. To address the AMR pandemic, there is an urgent need for rapid diagnostics that can determine if a patient has an infection, distinguish viral and bacterial infections, and identify the etiologic agent and its susceptibility profile (if bacterial or fungal). Much like for COVID-19, each individual in the clinical laboratory plays a role in fighting global AMR. Activities to ensure appropriate use of antimicrobials can occur at the bench, laboratory, institutional and regional level. Unlike COVID-19, which is caused by a single virus, SARS-CoV-2, AMR spans across a variety of microorganisms and affects all antimicrobials. Furthermore, continual evolution of resistance mechanisms, changes in organism prevalence, and introduction of new antimicrobials require continual adaptation. Suggested activities for the laboratory to ensure preparedness for this ongoing AMR pandemic are listed in Table 1.
Table 1. Ongoing “key” activities for clinical microbiology laboratories to ensure preparedness for the AMR pandemic

Select Resources for Table
CLSI. Performance Standards for Antimicrobial Susceptibility Testing. 30th ed. CLSI supplement M100. Wayne, PA: Clinical and Laboratory Standards Institute; 2020.
CLSI. Performance Standards for Antimicrobial Disk Susceptibility Tests. 13th ed. CLSI standard M02. Wayne, PA: Clinical and Laboratory Standards Institute; 2018.
CLSI. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically. 11th ed. CLSI standard M07. Wayne, PA: Clinical and Laboratory Standards Institute; 2018.
CLSI. Methods for Antimicrobial Dilution and Disk Susceptibility Testing of Infrequently Isolated or Fastidious Bacteria. 3rd ed. CLSI guideline M45. Wayne, PA: Clinical and Laboratory Standards Institute; 2016.
CLSI. Verification of Commercial Microbial Identification and Antimicrobial Susceptibility Testing Systems. 1st ed. CLSI guideline M52. Wayne, PA; Clinical and Laboratory Standards Institute; 2015.
For additional CLSI documents go here.
Publications and Websites
Messacar K, Parker SK, Todd JK, Dominguez SR. Implementation of rapid molecular infectious disease diagnostics: the role of diagnostic and antimicrobial stewardship. J Clin Microbiol. 2017;55(3):715-723. DOI: 10.1128/JCM.02264-16
Morency-Potvin P, Schwartz DN, Weinstein RA. Antimicrobial stewardship: how the microbiology laboratory can right the ship. Clin Microbiol Rev. 2017;30(1):381-407. DOI: 10.1128/CMR.00066-16.
FDA Susceptibility Test Interpretive Criteria (STIC) website
https://www.fda.gov/drugs/development-resources/fda-recognized-antimicrobial-susceptibility-test-interpretive-criteria. Accessed July 13, 2020.
CDC/FDA Antimicrobial Resistance (AR) Bank for isolates (can be used for verification studies)
https://www.cdc.gov/drugresistance/resistance-bank/index.html. Accessed July 13, 2020.
This article came from AST News Update, Volume 5, Issue 2 – July 2020 which is produced by the CLSI Outreach Working Group (ORWG). The ORWG is part of the CLSI Subcommittee on Antimicrobial Susceptibility Testing (AST) and was established in 2015. The formation of the working group originated in a desire to efficiently convey information regarding contemporary AST practices, recommendations, and resources to the clinical microbiology community. They welcome suggestions from you about any aspect of CLSI documents, educational materials, or their Newsletters.
